

Neurons (nerve cells) in the brain and central nervous system transmit signals to each other across connections called synapses. Glutamate is the primary neurotransmitter (messenger) that nerve cells use to send signals across these synapses to induce action in the brain. Glutamate enables the brain to develop and language to be learned. Without synapses that allow the chemical signal's transmission from one nerve cell to the next, nerve cells will not be able to communicate with each other. Other neurotransmitters carry inhibitory signals to reduce activity in the brain.
My research has shown that the post-synaptic density protein (PSD-95) stimulates the formation and maturing of the synapses that release glutamate, and increases the release of this neurotransmitter. Members of the PSD-95 family are involved in the development and organization of receptors that are clustered on the receiving side of the synapse. I am investigating how PSD-95 proteins regulate receptor clustering at synapses. This research is important because the number of receptors regulates the strength of the message: the more receptors, the stronger the message.
We want to gain a better understanding of how receptors accumulate at synapses, and how changes in this process may underlie long-term changes in synapse structure and function associated with learning and memory. If we can determine how to change the number of receptors, we can permanently enhance the signals received in the brain, which could improve learning and memory function. Also, by understanding how synapses are formed and how neurotransmitter receptor clustering is regulated, we may figure out how to rescue abnormalities in synapse formation and function associated with several neurological diseases such as Alzheimer's, mental retardation, schizophrenia and epilepsy.
Mechanisms that control synapse development
Brain function relies on communication between nerve cells through highly specialized junctions known as synapses where one neuron can release a transmitter to pass on a signal to the another cell. In humans, the majority of synapses form during early prenatal and postnatal development until about 1 year after birth. By the end of this period, the 100 billion neurons in the brain have each formed thousands of synapses. However, it is unclear how an arriving axon selects a particular postsynaptic partner, how pre- and postsynaptic proteins are recruited to the initial site of contact to form a functional synapse, and how synapse stability is maintained. By using molecular tools to identify and manipulate components of the synaptic machinery, research in the laboratory is focused on elucidating mechanisms that underlie the establishment and maturation of synapses.
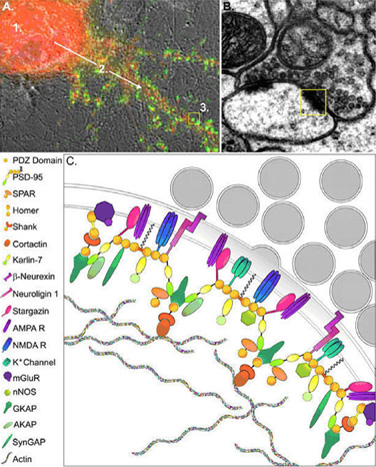
Protein Complexes at Excitatory Synapses. (A) An image of a neuron stained with the presynaptic marker synaptophysin (green), to identify synaptic contacts. This panel illustrates steps involved in the assembly of proteins at contact sites. Synapse formation is generally thought to involve three basic steps which include production of proteins in the cell soma (A-1), transport of these proteins to early sites of contact between axons and dendrites (A-2), and assembly of protein complexes at synapses (A-3). (B, C) The intense clustering of proteins seen at the PSD of excitatory synapses is highlighted in the electron micrograph shown in B. A schematic diagram of this region is blown up in C, illustrating the role of scaffolding molecules such as PSD-95 in assembly of large protein complexes. PSD-95 forms the core of the protein network, which is associated with the membrane through palmitoylation, and anchored within the postsynaptic compartment by several proteins that associates with actin. Coupling of PSD-95 to adhesion molecules such as neuroligins allows for trans-synaptic signaling.